Informed Consent for Research
Writing service by Research Data Experts in quantitative and qualitative research.
Our research data experts understand how to write informed consent for research participants. The experts recognize that informed consent should be clear and concise. The readability of informed consent is essential, which results in our Research Experts ensuring that the report has a Flesch Kincaid of 9.0 or less. The importance of informed consent makes it essential for the form to be written in a language so that potential participants can comprehend what you are asking them. For this reason, our informed consent writing services are provided by research writers who use simple language. The recommendation is that an informed consent form should be written below the 9th-grade reading level. Readability is standard practice for IRBs. When you order our services, we ensure that your informed consent will have a Flesch Kincaid score of 9.0 or less to support readability. Hence, when delivering your ordered informed consent writing services, we will attach a Flesch Kincaid score screenshot.
Required Elements of Informed Consent for Research
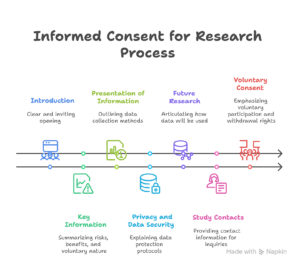
Different institutions have varied informed consent for research. However, in this section we focus on the commonly required elements of informed consent for research, which are introduction, key information, presentation of information collected, privacy and data security, future research, study contacts, and voluntary consent.
Introduction
The introduction of informed consent for a research study, paper, or project, whether quantitative, qualitative, or mixed methods can contain the title, and purpose of the study (3 to 4 sentences, and should be clear, straightforward, and devoid of research jargon), and researchers’ introduction. In the case the researcher is a doctoral learner, an example of an introduction is: I am [insert your name], a doctoral student under the supervision of Dr. [insert name of your dissertation Chair] in the College of Doctoral Studies [or if different, insert your specific College] at ABC University.
Key Information of an Informed Consent for Research Project
The focus of this section is helping potential research respondents or participants understand whether they are eligible to participate. Our research data experts fill this section of your informed consent will bullet points of the inclusion and exclusion criteria.
The other key piece of information is outlining the research activities, in particular, what participants are being asked to do. This section is important in assisting participants know what they are agreeing to in terms of time commitment and involvement.
Explaining when the activities will take place, where they will be conducted, and how they will be performed adds to the informed consent. We ensure to effectively explain who will have access to the data/information collected, the voluntary nature of the study, and any possible risks or discomfort that participants might experience. A statement of any direct benefit for research participants and paid compensation or incentives respondents might be eligible for their time. Is also included
Presentation of Information Collected Informed Consent for Research Interview, Research Survey or Research Questionnaire
Explaining where and how data will be presented once collected and analyzed adds to participants’ understanding. The description of the interview responses will be quoted while survey and questionnaire responses will be presented in aggregate, which is essential, together with the reassurance to participants that data will not be presented in a manner that can be used to de-identify them.
Privacy and Data Security of Informed Consent for Research Respondents
Reassuring privacy and data security is essential so that research respondents understand whether other researchers will be able to link data/responses back to them. Explaining to participants that no personally identifiable data will be collected or used and manner data will be stored securely is essential in improving participants’ willingness to be part of the study.
Future Research Section of Informed Consent for Research for Qualitative and Quantitative Research
It is important to inform participants that their de-identified data could be used in future research without additional consent.
Study Contacts Section Informed Consent for Quantitative and Qualitative Research
Including the principal investigator’s, co-investigators, and IRB chair’s contact information is important. Participants can use contact information to seek additional information, raise concerns, or report in the instances where they are placed at risk.
Voluntary Consent Section for Qualitative and Quantitative Research
The voluntary consent for qualitative and quantitative research outlines participants’ rights and requests to sign and issue the respective date if they agree to be part of the study or project.
Informed Consent for Research What to Expect from Our Writing Services
Get the informed consent research writing service from research data experts who understand that informed consent stands as the bedrock of ethical human subject research.
